UV water treatment system for pharma WFI India is now a legal requirement for every licensed pharmaceutical manufacturing facility under the Revised Schedule M 2025. The Central Drugs Standard Control Organisation has initiated immediate inspections across all pharma plants in India from January 2026.
Only 10 to 12 percent of manufacturers have applied for a compliance extension — meaning the vast majority of Indian pharma plants must comply with Revised Schedule M water system requirements right now. If your pharmaceutical plant does not have a validated UV water treatment system installed after your Reverse Osmosis unit, your facility is at serious risk of a CDSCO compliance notice, batch rejection or manufacturing licence suspension.
Alpha UV System, based in Noida, has been manufacturing CE and ISO certified UV water treatment systems for pharmaceutical WFI and Purified Water applications for 11 years. We have supplied validated UV systems to 200 plus pharma plants across India. This complete guide covers everything your quality team, plant head and purchase manager needs to know.
Pharma plants across India are now urgently upgrading their UV water treatment system for pharma WFI India compliance ahead of the January 2026 CDSCO inspection deadline.
What is WFI and Why UV Treatment is Mandatory Under Schedule M 2025
Water for Injection, commonly known as WFI, is the highest purity grade of water used in pharmaceutical manufacturing. It is used as a solvent for injectable drugs, for cleaning pharmaceutical equipment and vessels that contact sterile products, and as a component of sterile pharmaceutical formulations including IV fluids, ophthalmic solutions and injectable antibiotics.
The quality of WFI is governed in India by the Indian Pharmacopoeia, which specifies that WFI must contain less than 0.25 EU per ml of bacterial endotoxins, less than 100 colony forming units per ml of aerobic microbial count for purified water with WFI having no detectable microbial count, and a total organic carbon content of less than 500 ppb.
Achieving these standards consistently in a pharmaceutical manufacturing environment requires a multi-stage water treatment system. The standard pharmaceutical water treatment train in India includes raw water pre-treatment, softening, Reverse Osmosis, UV water treatment as the mandatory disinfection step, electrodeionisation or mixed bed polishing, and WFI storage and distribution loop.
The UV water treatment step is mandatory because RO membranes do not provide sterile filtration. Bacteria with dimensions smaller than the RO membrane pore size can pass through, and bacteria regrow rapidly in the post-RO storage tank if no disinfection step is present.
Under the Revised Schedule M 2025, CDSCO has strengthened water system requirements significantly. The DCGI has instructed State Licensing Authorities to initiate inspections based on the Revised Schedule M with immediate effect for all manufacturers who have not applied for an extension. This means your UV water treatment system will be examined during CDSCO inspections occurring right now across India in 2025 and 2026.
How UV Water Treatment Works in a Pharma Plant
UV water treatment uses ultraviolet light at a wavelength of 254 nanometers to destroy the DNA and RNA of microorganisms present in water. At this specific wavelength, UV photons are absorbed by the nucleic acids of bacteria, viruses and protozoa, forming thymine dimers that prevent the organism from replicating its genetic material. The organism becomes permanently inactivated — it cannot reproduce and is rendered completely harmless.
A correctly installed and validated UV water treatment system for pharma WFI India eliminates all bacteria, viruses and endotoxin-producing organisms before water enters the purified water storage tank.
Key Advantages of UV Treatment for Pharmaceutical Water Systems
- No chemical residue — UV treatment leaves absolutely no chemical residue in treated water. This is critical for pharmaceutical water systems where even trace levels of disinfection chemicals are unacceptable in purified water and WFI.
- No change in water chemistry — UV treatment does not alter pH, conductivity, ionic composition or TOC level. UV does not interfere with any downstream water quality parameters measured for pharmacopoeia compliance.
- Validated performance — UV systems can be validated to deliver a precise measured UV dose at a specified flow rate. This validation is documented in a UV dose validation certificate usable as compliance evidence during CDSCO and WHO GMP audits.
- Instant treatment — UV treatment works instantly. The water is fully disinfected as it passes through the UV chamber in seconds, unlike chemical disinfection which requires contact time of minutes to hours.
In a pharmaceutical plant, UV treatment units are typically installed at two critical points. The primary UV installation is at the post-RO pre-storage position — immediately after the RO unit and before the purified water storage tank. The secondary UV installation is within the WFI or purified water distribution loop, providing continuous recirculating treatment to prevent biofilm formation inside distribution piping.
Revised Schedule M 2025 — What Changed for Water Systems
Every pharmaceutical plant looking for a UV water treatment system for pharma WFI India must ensure their system comes with complete IQ OQ PQ documentation under Revised Schedule M 2025.
The Revised Schedule M 2025 represents the most significant update to Indian pharmaceutical GMP requirements in over two decades. The New Schedule M is a comprehensive framework introduced by CDSCO aimed at aligning India’s pharmaceutical manufacturing practices with international standards including WHO-GMP, encompassing facility design, equipment qualification, process validation, documentation and quality management systems.
Critical Changes for Water Systems Under Revised Schedule M 2025
- Mandatory qualification of water systems — The revised schedule explicitly requires Installation Qualification, Operational Qualification and Performance Qualification for all pharmaceutical water systems including UV treatment units. Previously many smaller pharma plants operated UV systems without formal qualification documentation.
- Alignment with WHO GMP water system requirements — The revised schedule brings Indian water system requirements in line with WHO Technical Report Series No. 970 on pharmaceutical water systems, including specific requirements for UV dose validation, distribution loop design and microbiological monitoring frequency.
- Stricter documentation requirements — Pharma plants must now maintain calibration records for UV intensity meters, UV lamp replacement records with hours of operation, and periodic microbiological test results at defined sampling points in the water distribution system.
- Risk-based approach to water system monitoring — The new schedule emphasises identifying potential risks at various stages of production and implementing appropriate controls. For water systems this means identifying and documenting the risk of UV system failure and having a validated response protocol.
Under Revised Schedule M 2025, pharma plants purchasing a UV water treatment system for pharma WFI India must ensure it comes with complete validation documentation including IQ OQ PQ protocols, a UV intensity monitor for continuous dose verification, and a lamp hour counter for maintenance scheduling. Alpha UV System provides all these as standard with every pharma UV installation.
UV Dose Requirements for Different Pharma Water Applications
Selecting the correct UV dose for your specific pharmaceutical water application is critical. An undersized UV system delivering insufficient UV dose at peak flow rate will fail to inactivate all microorganisms, creating a hidden compliance risk that may not be discovered until a CDSCO audit or batch rejection event.
Alpha UV System sizes every UV water treatment system for pharma WFI India to deliver the validated dose at peak flow
rate, not average flow rate, ensuring consistent compliance during CDSCO audits.
| Application | UV Dose | Reason |
| Purified Water — post-RO storage tank inlet | 40 mJ/cm² | 4-log reduction of standard bacteria including E. coli and Pseudomonas |
| WFI Distribution Loop — continuous treatment | 60 mJ/cm² | Higher dose accounts for radiation-resistant organisms in critical WFI systems |
| WFI at Point of Use | 80 – 100 mJ/cm² | Systems with dead legs or reduced flow velocity areas |
| HPWFI — Highly Purified Water for Injection | 100 – 120 mJ/cm² | EU GMP Annex 1 aligned pharma plants |
| Biofilm Elimination Flush Treatment | 150+ mJ/cm² | Combined with hot water or ozone in validated sanitisation protocol |
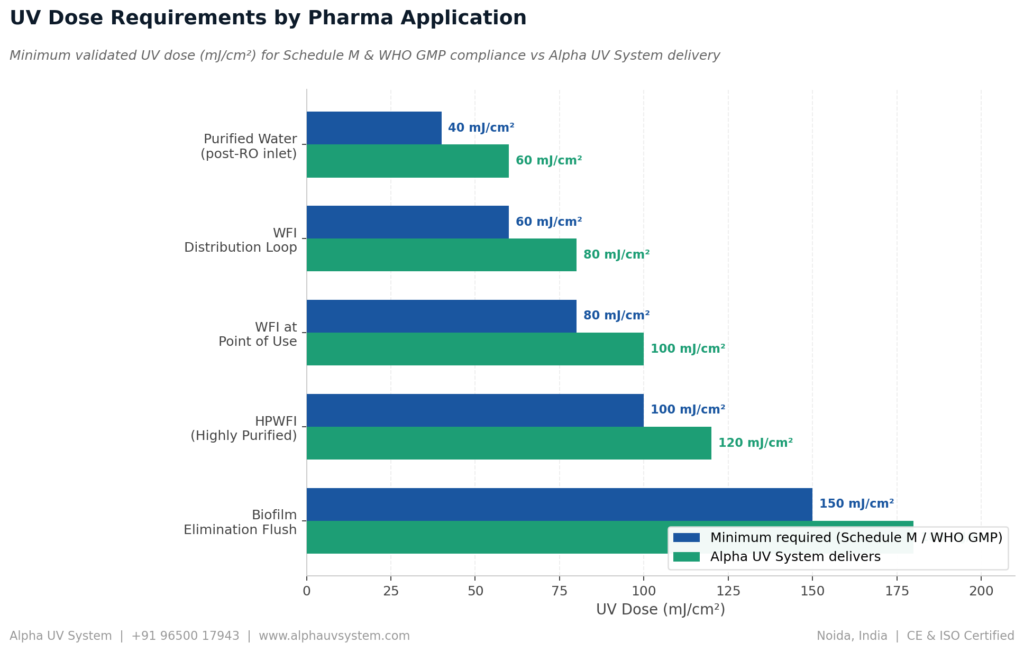
Alpha UV System calculates the UV dose delivered at maximum flow rate for every pharma UV system we manufacture. This calculation is verified using a NABL-calibrated UV intensity meter during factory acceptance testing and documented in the UV dose validation certificate supplied with the system.
Correct Installation Sequence — Where to Place the UV System
The position of the UV treatment unit in the pharmaceutical water treatment train is critical. An incorrectly positioned UV system will not achieve pharmacopoeia-compliant water quality even if it is correctly sized and functioning properly.
Correct Pharmaceutical Water Treatment Sequence
- Raw water inlet — borewell, municipal or surface water
- Multimedia sediment filter — removes suspended particles above 10 microns
- Activated carbon filter — removes chlorine, chloramines and organic compounds
- Antiscalant dosing — prevents RO membrane scaling
- Reverse Osmosis membrane — removes dissolved solids, heavy metals, reduces bacterial load
- UV water treatment unit at 254nm — destroys residual bacteria — the mandatory Schedule M step
- Electrodeionisation or Mixed Bed — achieves ultra-low conductivity for WFI grade
- 0.2 micron sterilising filter — final particulate barrier before storage
- Purified Water or WFI storage tank — SS 316L with sanitary fittings
- Distribution loop with continuous circulation and loop UV treatment
| CRITICAL RULE: The UV system must always be installed AFTER the RO membrane, not before it. Installing UV before RO is the most dangerous installation mistake in pharmaceutical water systems. The RO membrane surface harbours bacteria. If UV is installed before RO, the bacteria on the membrane will contaminate water after it has been UV treated, defeating the entire purpose of the UV unit. |
Schedule M Compliance — What CDSCO Auditors Check for UV Systems
When CDSCO auditors inspect a pharmaceutical facility under Revised Schedule M 2025, the UV water treatment system is examined systematically against qualification and maintenance documentation requirements. Understanding exactly what auditors check helps pharma QA teams prepare documentation in advance.
During a CDSCO inspection, auditors specifically verify that the UV water treatment system for pharma WFI India has been qualified with documented IQ, OQ and PQ protocols.
Documentation CDSCO Auditors Examine
- Installation Qualification report — confirms UV system was installed according to manufacturer specifications, all components correctly identified, all utility connections meet GMP requirements
- Operational Qualification report — confirms UV system delivers specified UV dose at designed flow rate, verified by UV intensity measurement with a calibrated UV meter
- Performance Qualification report — confirms water downstream of UV system meets pharmacopoeia microbiological limits under actual production conditions over minimum 30 days
Physical Inspection of the UV System
- UV lamp operational status — confirmed by UV lamp indicator light or monitoring system
- Quartz sleeve condition — physically inspected for scale or fouling. A fouled sleeve is visible evidence of inadequate maintenance and will result in a GMP finding
- UV intensity monitor — auditors confirm a UV intensity monitor is installed and providing continuous dose verification
- Lamp hour counter — checked to confirm lamp has not exceeded its rated 9000 hours. Exceeded lamp hours is a direct compliance finding
Records That Must Be Available on Request
- Lamp replacement log with dates and hours of operation
- UV intensity readings taken at defined intervals using a calibrated meter
- Quartz sleeve cleaning records with dates and cleaning agent used
- Calibration records for the UV intensity meter used for monitoring
- Microbiological test results from sampling points downstream of the UV system
Alpha UV System provides a complete CDSCO audit-ready documentation package with every pharma UV installation. Our IIT Patna-educated team is available to support pharma plants during pre-audit preparation at no additional charge.
WHO GMP Requirements for UV Water Treatment India
Indian pharmaceutical companies exporting formulations to regulated markets — USA, Europe, UK, Australia, Japan — must maintain WHO GMP or US FDA compliance in addition to domestic Schedule M requirements. UV water treatment system requirements under these international standards are broadly aligned with Revised Schedule M 2025 but include some additional specifications.
WHO Technical Report Series No. 970 on pharmaceutical water systems specifies that UV treatment systems used in pharmaceutical water systems must be validated to deliver a minimum UV dose of 40 mJ per square centimetre for purified water and 60 mJ per square centimetre for WFI. The WHO guidelines also specify that UV systems must be equipped with a UV intensity monitor that provides continuous verification of the delivered UV dose during production.
UV systems for pharmaceutical water must be validated for dose delivery, equipped with UV intensity monitors, and designed for easy lamp replacement and quartz sleeve cleaning. The NSF/ANSI 55 standard provides a framework for UV system validation, though pharmaceutical applications typically exceed the minimum requirements of this standard.
Alpha UV System has supplied UV water treatment systems to pharmaceutical companies operating under WHO GMP certification in Baddi, Haridwar, Hyderabad and Ahmedabad. Our validation documentation is accepted by WHO GMP auditors without modification. For pharma companies targeting US FDA compliance, our systems meet 21 CFR Part 211 equipment qualification requirements.
Five Most Common UV System Installation Mistakes in Pharma Plants
After 11 years of installing and auditing UV water treatment systems in pharmaceutical plants across India, our engineering team has identified five installation mistakes that repeatedly appear during CDSCO compliance failures.
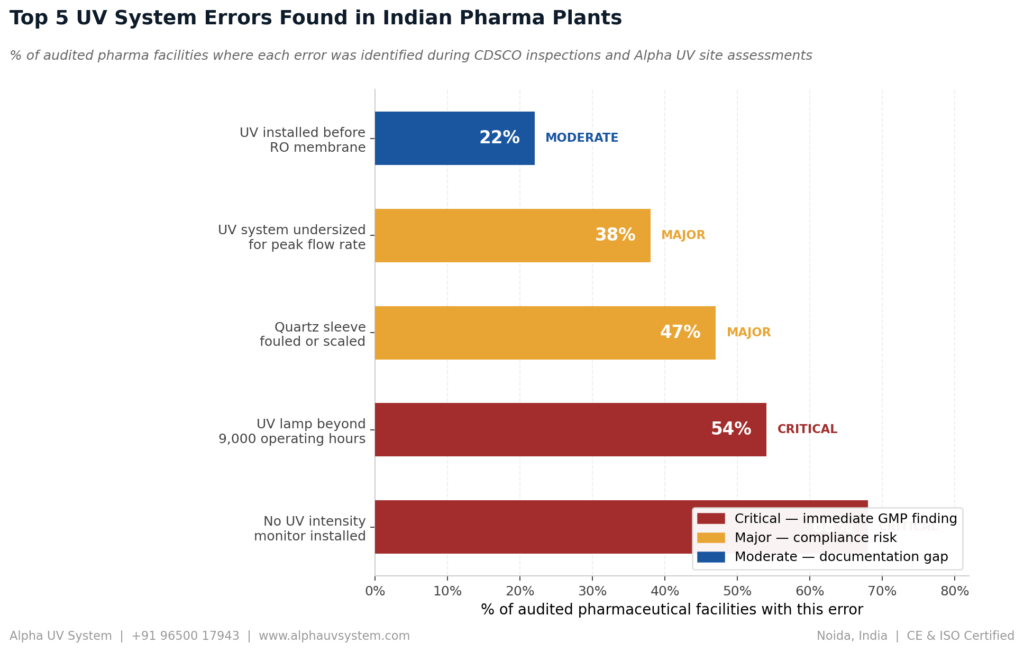
Mistake 1 — UV Installed Before RO Membrane
This is the most dangerous and most common mistake. The RO membrane surface harbours bacteria. Installing UV before the RO unit allows RO membrane bacteria to contaminate already-treated water. Always install UV after RO without exception.
Mistake 2 — UV System Undersized for Peak Flow Rate
Many pharma plants calculate UV system size based on average daily water consumption. A UV system delivers the correct UV dose only when actual flow rate does not exceed its rated maximum. If actual peak flow rate exceeds the UV system rated maximum, the UV dose drops below the required level — potentially below the threshold for complete bacterial inactivation. Alpha UV System sizes every UV system for the maximum possible flow rate, not the average.
Mistake 3 — Quartz Sleeve Not Cleaned Regularly
The quartz sleeve surrounding the UV lamp is the most maintenance-critical component. Even moderately hard water will deposit calcium scale on the quartz sleeve within 3 to 6 months of operation. This scale significantly reduces UV transmission, reducing the actual UV dose delivered to the water even though the lamp appears to be functioning normally. Quarterly inspection and 6-monthly cleaning with 10 percent citric acid solution is essential.
Mistake 4 — UV Lamp Not Replaced at 9000 Hours
UV lamps lose germicidal output at 254 nanometres long before they lose visible light output. A UV lamp operating beyond 9000 hours may appear to be functioning normally but is delivering significantly less germicidal UV dose than required. CDSCO auditors specifically check lamp hour counters. If the lamp has exceeded 9000 hours, it is a compliance finding regardless of whether the water is passing microbiological tests.
Mistake 5 — No UV Intensity Monitor Installed
Revised Schedule M 2025 requires continuous verification of UV system performance. Operating a UV system without a UV intensity monitor means there is no evidence that the system was delivering the required UV dose during the period between audits. Even if the water was microbiologically compliant, the absence of continuous UV dose monitoring data is itself a GMP compliance finding.
Maintenance Schedule for Pharma UV Systems
A compliant pharmaceutical UV water treatment system requires four maintenance activities performed on a defined schedule. This schedule should be documented in the Standard Operating Procedure and records of each maintenance activity filed in the UV system qualification file.
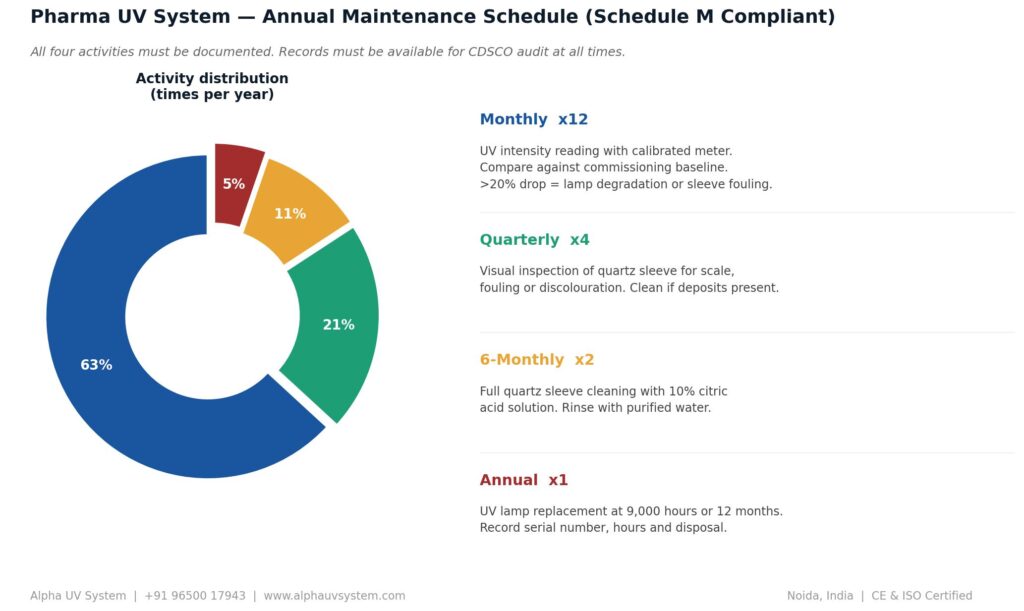
Monthly — UV Intensity Reading
Measure and record the UV intensity at the quartz sleeve using a calibrated UV intensity meter. Compare against the original commissioning baseline. A reduction of more than 20 percent from baseline indicates fouling or lamp degradation requiring investigation.
Quarterly — Quartz Sleeve Visual Inspection
Open the UV chamber and visually inspect the quartz sleeve for scale, fouling or discolouration. If scale is visible, clean with 10 percent citric acid solution, rinse with purified water and dry before reassembly.
Six-Monthly — Quartz Sleeve Cleaning
Regardless of visual appearance, clean the quartz sleeve with citric acid solution every six months as a preventive maintenance measure to maintain maximum UV transmittance.
Annual — UV Lamp Replacement
Replace the UV lamp every 12 months or at 9000 hours of operation, whichever comes first. Record the old lamp’s hours of operation, disposal details and the new lamp’s serial number and installation date. Alpha UV System supplies genuine Philips UV lamps — the global standard for germicidal UV lamp performance with guaranteed 12,000-hour rated life.
Why 200+ Pharma Plants in India Choose Alpha UV System
When sourcing a UV water treatment system for pharma WFI India, the three non-negotiable criteria are CE certification, validated UV dose documentation and a supplier with pharmaceutical water system expertise.
IIT Patna Educated Engineering Team
Our team includes MBA graduates from IIT Patna — India’s Institute of National Importance. This technical education level means our team speaks the language of pharmaceutical quality — validation protocols, IQ OQ PQ documentation, GMP compliance, risk assessment — fluently and accurately. When your QA manager calls us with a question about UV dose validation for a WHO GMP audit, they get a technically accurate answer within two hours, not a sales call.
Complete Validation Documentation as Standard
Every Alpha UV System pharma UV installation includes a complete documentation package at no additional charge: UV system specifications, installation procedure, UV dose validation certificate with actual measured UV intensity data, IQ OQ PQ protocol templates, calibration record for the UV intensity meter used for validation, and a CDSCO audit-ready SOP for UV system operation.
Factory-Direct Pricing 30 to 40 Percent Below European Brands
European UV system brands have dominated the Indian pharmaceutical market for decades. Their products are technically excellent but priced for European markets and carry distributor margins making them 30 to 40 percent more expensive than Alpha UV System for equivalent technical performance. Our systems are CE certified — the same standard as European equipment — and our validation documentation is accepted by the same CDSCO and WHO GMP auditors who accept European brand documentation.
Trusted by pharma plants in Baddi, Haridwar, Hyderabad and Ahmedabad. 200 plus client installations. 11 years manufacturing experience. Zero recalls. Zero regulatory actions arising from UV system non-compliance.
Technical Specifications — Alpha UV System Pharma Range
| Specification | Detail |
| Chamber Construction | SS 316L electropolished, sanitary end connections, zero dead leg design for WFI loop applications |
| UV Lamp Type | Low pressure, high output at 254 nanometres — genuine Philips germicidal UV lamps |
| UV Lamp Wattage | 16W to 120W depending on flow rate and required UV dose |
| Flow Rate Range | 200 LPH to unlimited LPH — any pharmaceutical plant capacity accommodated |
| UV Dose Range | 40 mJ/cm² to 150 mJ/cm² depending on application |
| UV Intensity Monitor | Digital display, 4-20 mA output for BMS/SCADA integration, alarm output for low UV intensity |
| Quartz Sleeve | High-purity synthetic quartz, transmittance greater than 90% at 254 nanometres |
| Connections | Tri-clamp sanitary fittings standard for pharma — flanged connections available |
| Certifications | CE, ISO 9001, RoHS, UL |
| Validation Documentation | UV dose validation certificate, IQ OQ PQ protocol templates, calibration records, SOP |
| Warranty | 12 months from date of commissioning on all components except UV lamp |
View our complete UV Water Treatment System range at our page
Frequently Asked Questions
Q: Is UV water treatment mandatory for pharma manufacturing in India under Revised Schedule M 2025?
A: Yes. The Revised Schedule M 2025 requires validated water systems for all pharmaceutical manufacturing facilities. UV treatment is the pharmacopoeia-accepted method for microbial control in purified water and WFI systems. CDSCO inspections under Revised Schedule M 2025 are being conducted from January 2026 and specifically examine water system qualification documentation.
Q: What UV dose is required for pharma purified water?
A: A minimum of 40 mJ per square centimetre at the maximum design flow rate for purified water storage tank inlet. For WFI distribution loop treatment, 60 mJ per square centimetre is recommended. Alpha UV System validates all pharma UV systems to the correct dose for the specific application.
Q: How often must pharma UV lamps be replaced?
A: Every 9000 hours of operation or 12 months, whichever comes first. This is a specific compliance requirement under Revised Schedule M 2025. Exceeding 9000 hours without lamp replacement is a GMP finding during CDSCO audits.
Q: Does Alpha UV System provide IQ OQ PQ validation documentation?
A: Yes. Complete IQ, OQ and PQ protocol templates plus a UV dose validation certificate with actual measured UV intensity data are provided with every Alpha UV System pharma UV installation at no additional charge.
Q: What is the UV water treatment system capacity range available?
A: Alpha UV System manufactures UV water treatment systems for pharma WFI India from 200 LPH to unlimited LPH. Any pharmaceutical plant flow rate can be accommodated — from small 200 LPH purified water systems to high-volume WFI systems for large multi-product injectable plants.
Q: Is Alpha UV System CE certified?
A: Yes. All Alpha UV System UV water treatment systems are CE certified, ISO 9001 certified, RoHS certified and UL certified. CE certification means our systems meet European safety standards and are accepted by WHO GMP and EU GMP auditors operating in India.
Q: Where can I get a UV water treatment system for pharma WFI India with same-day quotation?
A: Alpha UV System provides factory-direct quotations within 2 hours of receiving your flow rate and application details. Call +91 96500 17943 or visit www.alphauvsystem.com. Our IIT Patna-educated team responds within 2 hours on all working days.
Q: What is the best UV water treatment system for pharma WFI India?
A: The best UV water treatment system for pharma WFI India is one that delivers a validated UV dose of minimum 40 mJ per square centimeter at maximum flow rate, uses SS 316L chamber construction, provides complete IQ OQ PQ documentation and is CE certified. Alpha UV System meets all four criteria and provides factory-direct pricing 30 to 40 percent below European brands. Call +91 96500 17943.
Learn more about our pharma UV water treatment solutions
on our Pharma Water Treatment page.| About Alpha UV System Alpha UV System is a CE and ISO certified UV water treatment system manufacturer based in Noida, Uttar Pradesh, India. Established under the OZ-India Group alongside Ozone India Technology and Oz Mist Food Technology, Alpha UV System has 11 years of manufacturing experience and 200 plus client installations across pharmaceutical, food processing, hospital, STP, swimming pool and industrial applications across India. Our engineering team includes MBA graduates from IIT Patna — India’s Institute of National Importance. We manufacture UV water treatment systems from 200 LPH to unlimited LPH. CE certified, ISO 9001 certified, RoHS and UL certified. Authorised Philips UV lamp distributor. Trusted by Bisleri, Taj Hotels, IIT, Britannia, Coca-Cola, Parle Agro, Hyundai, Tata and McDonald’s. Exporting to UAE, Kenya and Bangladesh. For the most trusted UV water treatment system for pharma WFI India, Call: +91 96500 17943 | Website: www.alphauvsystem.com | Noida, Uttar Pradesh, India |
External Links
• WHO Guidelines for Pharmaceutical Water Quality — World Health Organization
• CDSCO Revised Schedule M 2025 Official Notifications — CDSCO India

